TilS
Lysidine, a lysine-combined modified cytidine, is exclusively located at the anticodon wobble position (position 34) of eubacterial tRNA(Ile)(2) and not only converts the codon specificity from AUG to AUA, but also converts the aminoacylation specificity from recognition by methionyl-tRNA synthetase to that by isoleucyl-tRNA synthetase (IleRS).
 |
||
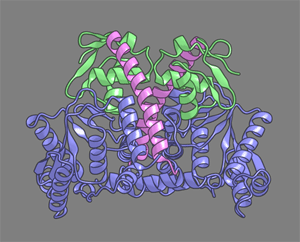
| Here, we report the crystal structure of lysidine synthetase (TilS) from Aquifex aeolicus at 2.42-A resolution. TilS forms a homodimer, and each subunit consists of the N-terminal dinucleotide-binding fold domain (NTD), with a characteristic central hole, and the C-terminal globular domain (CTD) connected by a long alpha-helical linker. | ||
The NTD shares striking structural similarity with the ATP-pyrophosphatase domain of GMP synthetase, which reminds us of the two-step reaction by TilS: adenylation of C34 and lysine attack on the C2 carbon. Conserved amino acid residues are clustered around the NTD central hole. Kinetic analyses of the conserved residues' mutants indicated that C34 of tRNA(Ile)(2) is adenylated by an ATP lying across the NTD central hole and that a lysine, which is activated at a loop appended to the NTD, nucleophilically attacks the C2 carbon from the rear.
 |
||
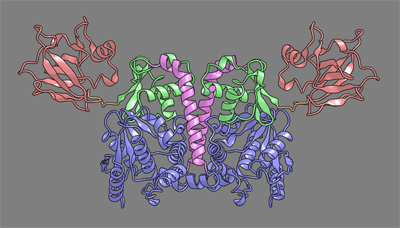
| Escherichia coli TilS (called MesJ) has an additional CTD, which may recognize the tRNA(Ile)(2) acceptor stem. In contrast, a mutational study revealed that A. aeolicus TilS does not recognize the tRNA acceptor stem but recognizes the C29.G41 base pair in the anticodon stem. Thus, the two TilS enzymes discriminate tRNA(Ile)(2) from tRNA(Met) by strategies similar to that used by IleRS, but in distinct manners. | ||
![[XXX]](/image/tw.png)