ArcTGT-tRNA complex
Transfer RNA (tRNA) canonically has the clover-leaf secondary structure with the acceptor, D, anticodon, and T arms, which are folded into the L-shaped tertiary structure. To strengthen the L form, posttranscriptional modifications occur on nucleotides buried within the core, but the modification enzymes are paradoxically inaccessible to them in the L form.
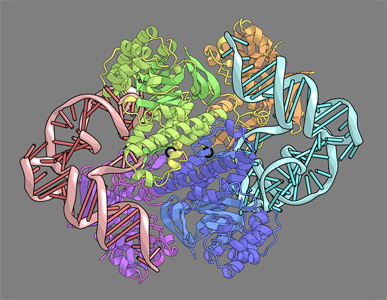 |
| In this study, we determined the crystal structure of tRNA bound with archaeosine tRNA-guanine transglycosylase, which modifies G15 of the D arm in the core.
|
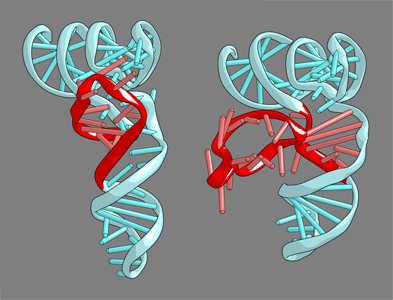 |
| The bound tRNA assumes an alternative conformation ("λ form") drastically different from the L form. All of the D-arm secondary base pairs and the canonical tertiary interactions are disrupted. Furthermore, a helical structure is reorganized, while the rest of the D arm is single stranded and protruded.
|
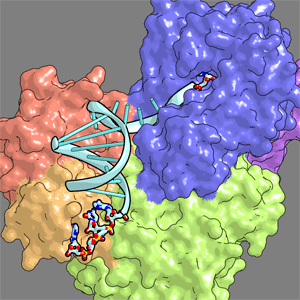 |
| Consequently, the enzyme precisely locates the exposed G15 in the active site, by counting the nucleotide number from G1 to G15 in the lambda form.
|
Gallery


![[XXX]](/image/tw.png)